Many homemade liqueurs seem perfectly balanced at first but taste softer months later. This shift is rarely caused by alcohol loss. Instead, slow chemical reactions such as sugar inversion, ester formation, alcohol integration, and mild oxidation reshape the flavor structure.
Sweetness can increase perceptually while acidity decreases, making alcohol feel less present. Understanding these processes helps you control balance, prevent over-sweetening, and adjust structure when needed.

On this page
Many home liqueur makers notice a strange shift after bottling. A fruit liqueur tastes balanced when it’s first finished: the alcohol feels supportive, the sweetness feels controlled, and the flavor has structure. Then, after a few months in storage, it starts to taste “watered down.”
What makes this confusing is that nothing obvious has happened. The fruit flavor is still present, the sweetness is still present, and there’s no sign of spoilage, fermentation, bubbling, haze, or pressure. Yet the alcoholic bite feels muted, as if the drink has been diluted.
This usually triggers a few big questions. Did alcohol evaporate through the bottle? Did something chemically remove ethanol from the mixture? Or was the liqueur oversweetened at the end, and time simply revealed it?
Liqueur Alchemy viewpoint: In most properly stored liqueurs, alcohol doesn’t vanish. The drink evolves, and your palate reads it differently as chemistry reshapes sweetness, acidity, aroma, and “heat.”
If your liqueur is stored in sealed glass and left unopened, ethanol doesn’t meaningfully evaporate. At around 20% ABV, you’re also outside the comfort zone for fermentation, because most yeasts fail well below that range and bacteria are generally inhibited.
So if nothing leaked and nothing was added, the alcohol percentage is effectively the same as the day it was bottled. What changes is not the ABV, but the perceived balance between alcohol warmth, fruit intensity, sugar sweetness, and acidity.
In other words, “watered down” is often a sensory interpretation, not a physical event.
Freshly blended liqueurs often taste sharper than they will later, especially if syrup was added close to the finish. Early on, ethanol can feel separate from the rest of the liquid, which makes the alcohol seem more obvious and more “present.”
Over weeks and months, ethanol integrates into the overall matrix of sugars, acids, and aromatic compounds. This doesn’t remove alcohol; it smooths the sensation of alcohol. The same ABV can feel less hot, less spiky, and less aggressive.
That softening is one reason aged spirits feel smoother than young spirits. Fruit liqueurs don’t age like whiskey in a barrel, but they do mellow. And when the burn fades, many people interpret the change as dilution.

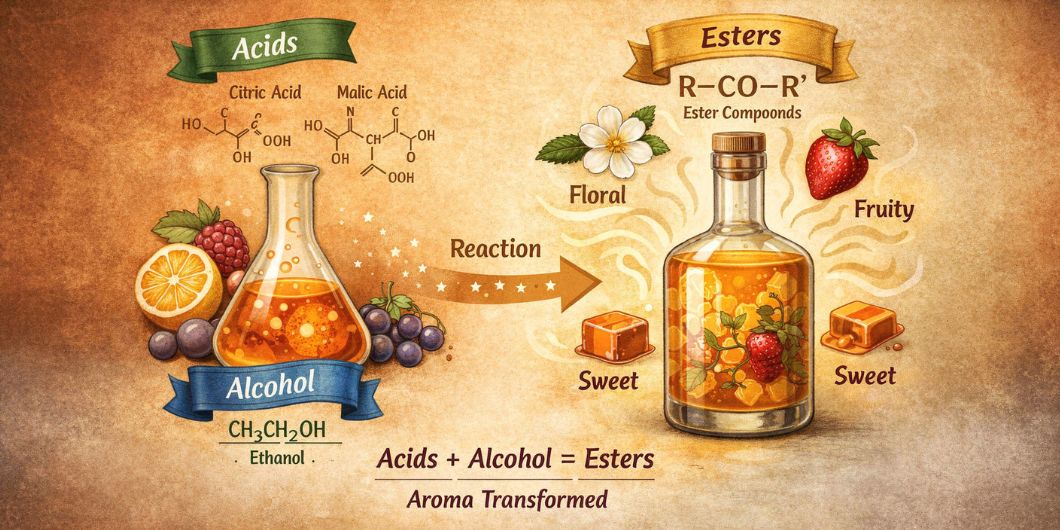
Fruit liqueurs are naturally acidic because fruit contains carboxylic acids such as citric, malic, or tartaric acid. That acidity doesn’t just affect taste — it changes sugar behavior over time.
In acidic conditions, sucrose can slowly break down into glucose and fructose. This process is called inversion, and it matters because fructose in particular tastes sweeter than sucrose. The liqueur can become perceptually sweeter even though you didn’t add more sugar.
As sweetness rises, alcohol warmth becomes less noticeable. Alcohol is easiest to detect when a drink is relatively dry and bright. Increase sweetness and soften acidity, and the alcohol recedes into the background.
Practical takeaway: Sweetness often “creeps forward” with time. That’s why sweetening to the edge right after straining can become too much a month later.
Alcohol and fruit acids can slowly react to form esters. In a lab this reaction is pushed with strong acids and heat. In a bottle at room temperature, it happens gradually over months.
Esters are powerfully aromatic. Many fruity, floral, candy-like, and creamy notes are ester-driven. As ester formation increases, the liqueur can smell more “fruit-forward” even as the palate feels smoother.
Two changes occur at the same time. Acidity is reduced as acids are converted, and aroma complexity increases as esters build. Lower acidity reduces sharpness, and higher aroma shifts attention away from ethanol warmth.
Even a closed bottle contains oxygen in the headspace. Over time, small oxidative reactions can subtly reshape the flavor. This isn’t spoilage — it’s slow, normal chemistry.
Oxidation can reduce bright top notes, soften some tannins or phenolics, and slightly flatten the “lift” that makes a liqueur feel vivid. Warm storage accelerates these changes, so a cupboard in a warm home will evolve faster than a cool cellar shelf.
Oxidation doesn’t remove alcohol, but it can reduce structure. When structure drops, alcohol feels less anchored and less noticeable.
One of the most common real-world reasons for “watered down” liqueur is simply this: it was sweetened a little too much right after the infusion was strained.
Early on, alcohol bite is sharper. That sharpness can make the drink feel harsher than it will later, encouraging you to add more syrup until it tastes comfortable. But once the liqueur rests, alcohol integrates and sugar inversion increases perceived sweetness.
Weeks later, you may end up with a liqueur that is sweeter than the alcohol can carry. The result isn’t truly diluted — it’s just unbalanced. Fruit and sweetness dominate, and ethanol support becomes quiet.
Rule of thumb: If you sweeten “perfectly” on day one, it can be too sweet by week six. Leave yourself room.
Both outcomes are possible, depending on the ingredients and the build. Some liqueurs improve dramatically with time because ester formation and integration create a smoother, richer profile. Classic examples include deeply colored fruit liqueurs and high-tannin berries.
Other liqueurs can lose brightness. Delicate fruits, fresh citrus, and very floral profiles may flatten or shift in a direction that feels less energetic. This isn’t wrong — it’s just evolution.
Whether aging helps depends on fruit type, sugar level, acidity, alcohol percentage, oxygen exposure, and storage temperature.
Yes. Blending is a legitimate method, and it’s often the simplest fix when a liqueur has become too sweet or too soft after resting.
The best approach is to add alcohol in small steps. A small increase can shift the balance quickly, and the mixture should be allowed to rest again so it reintegrates before you judge it.
Tip: If you can, write down batch volumes. Corrections are easier when you can repeat them.
Sometimes the liqueur doesn’t need more alcohol — it needs more structure. Structure is the contrast that keeps sweetness and fruit from feeling flat. It often comes from acidity, bitterness, tannin, and spice.
A small addition of acid (such as citric or malic, diluted and added carefully) can restore brightness. A bitter citrus tincture can bring back a firm finish. A short spice re-infusion can add backbone and reintroduce complexity.
The goal is not to make the liqueur harsher. The goal is to bring back tension so the alcohol feels supportive again.
What feels like dilution is usually evolution. Fruit acids convert. sugars shift in sweetness. esters form. oxidation softens edges. alcohol integrates. the sensory spotlight moves.
Homemade liqueurs are not static liquids. They’re living chemical systems that continue changing long after bottling, especially in warm storage.
The craft lesson is simple: measure when you can, sweeten conservatively, rest before final decisions, and accept that “smooth” can feel like “weaker” even when the ABV is unchanged.
Mastery mindset: Alcohol rarely disappears. Balance changes — and once you understand why, you can control it.
Can alcohol evaporate from a sealed bottle of liqueur?
Why does the alcohol “bite” disappear over time?
Does sugar become sweeter in fruit liqueurs as they age?
Does adding too much simple syrup cause liqueur to taste diluted later?
Can I add more alcohol to a liqueur after it has aged?
This website uses cookies to ensure you get the best experience on our website. Read our Privacy Policy.